Mending the Myths: Facts about Silicone Gel-filled Breast Implants
Published on April 21, 2011 by Brian Joseph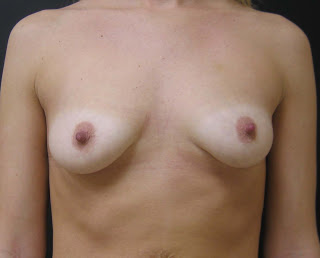
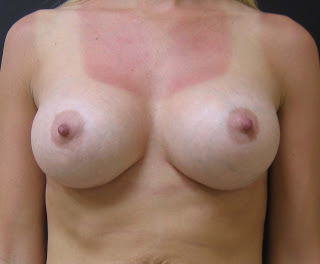
These are five myths about silicone implants addressed by the Allergan Pharmaceuticals’ website.
1. Safety of silicone products in the body. Silicone gel implants have been shown to be safely used in the body as well as for many different medical products and devices, which include joint replacement, heart valves, shunts, pacemakers and of course, silicone implants and silicone shell implants.
2. During routine mammography or exercising, it is significantly possible for silicone implants to rupture. This fact is a myth and according to Allergan Pharmaceuticals, it is very unlikely that silicone gel-filled implants would rupture during routine physical activity or a routine mammogram. The shells of the implants today are much stronger and thicker and “have been designed to withstand more than 25 times the force of a normal mammogram without failure.” Allergan notes that saline and silicone implants are not lifetime devices and it is possible that some point in the patients lifetime that the implants will need to be replaced or removed.
3. Silicone gel implants may cause autoimmune diseases or cancer. The actual facts are that silicone gel implants are highly studied medical devices with thousands and thousands of peer reviews that have been published with epidemiological studies supporting their safe use. The numerous studies have shown that there is no evidence concluding that silicone gel will increase the risk of cancer, autoimmune diseases like rheumatoid arthritis, lupus, or any neurological diseases. In fact, “The National Institute of Health’s Institute of Medicine (IQM) released a 400 page report in 1999 that was entitled Safety of Silicone Breast Implants, concluding that a review of toxicology studies of silicone and other substances known to be in breast implants does not provide a basis for health concern.”
4. Breast implants can grossly impact a mother’s ability to breastfeed or lactate. According to Allergan Pharmaceuticals, the 1999 IQM Report has concluded there is no evidence of increased levels of silicone in the breast milk for women who have implants and it is not harmful to the infants. In fact, the Committee strongly concluded that mothers with silicone implants should attempt breastfeeding and stated that the evidence for health effects in children related to maternal breast implants is insufficient.
5. Breast augmentation surgery may result in disproportionate or unnatural results. In general, women who undergo breast implant surgery have different views and different desires. In my practice at Park Avenue Plastic Surgery in Orlando Florida, I feel it is important to select implants that are in proportion to a patients figure and frame. I use a variety of different implant sizes, shapes and fillers (both silicone and saline), to achieve the desired size and appearance a patient desires.
Archives
- January 2025
- October 2024
- August 2024
- May 2024
- February 2024
- October 2023
- April 2020
- March 2020
- November 2019
- October 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- October 2016
- February 2016
- September 2015
- July 2015
- November 2014
- September 2013
- August 2013
- June 2012
- May 2012
- April 2012
- March 2012
- February 2012
- January 2012
- November 2011
- September 2011
- August 2011
- July 2011
- June 2011
- May 2011
- April 2011
- February 2011
- December 2010
- October 2010
- September 2010
- August 2010
- July 2010
- June 2010
- May 2010
- April 2010
- March 2010
- February 2010
- January 2010
- December 2009
- November 2009
- October 2009
- September 2009
- August 2009
- July 2009
- May 2009
- March 2009
- February 2009
- January 2009
- December 2008
- November 2008
